 
As energy is added, the randomness of the molecular motion increases. The entropy of a pure crystalline solid at absolute zero is zero. In the Table, (t) refers to crystal-crystal transitions (m) to crystal-liquid transitions (c), (1), and (g) to crystal, liquid, and gas G to glass transition and m, s to metastable, stable crystal. Table 2, containing results derived from measurements of heat capacities of some pure organic compounds published since 1961, gives values of temperatures, enthalpies, and entropies of phase transitions, and also of standard entropies. The standard state ionic entropies given in table 10 are corrected for. The formation values refer to the reaction Ln, Ln"a while the hydration values relate to the use of eqs. Values for yttrium hydration are also included when available. Tables 9-11 list the predicted thermodynamic functions for the hydration of divalent, trivalent and tetravalent lanthanides as calculated by Bratsch and Lagowski (1985b). 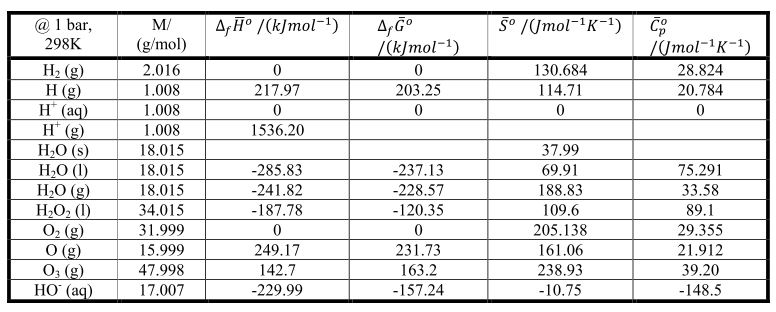
The data reported by various authors are recorded in Table 16.4. S, and specific heat capacity, C, for COCIF (ideal gas state, atmospheric pressure, 25 C) have been calculated from molecular data. The standard values of the enthalpy, H, entropy. If the temperature is not too far removed from 298-16 °K we may, to a first approximation, replace the heat and entropy terms in (10.51) by their standard values at 298 16° which may be calculated from the data in table 8.1. Note that the molar entropy thus calculated based on the third law of thermodynamics is occasionally called absolute entropy. A few of them will be listed as the standard molar entropy, s, in Table 5.1. In chemical handbooks we see the tabulated numerical values of the molar entropy calculated for a number of pure substances in the standard state at temperature 298 K and pressure 101.3 kPa. As shown in the figure, the molar entropy of a pure substance increases with increasing temperature. If a structural transformation occurs in the solid state, an additional increase in the molar entropy comes from the heat of the transformations. įigure 3.6 shows schematically the molar entropy of a pure substance as a function of temperature. The value for 298.15 K is calculated by the authors from a function fitted to the original data. Use those data to calculate the standard entropy of adenine at 298.15 K, which assume the Debye relationship for Cp. Table 11.8 contains a sampling of their data over the range from 7.404 K to 298.15 K. have measured the heat capacity of crystalline adenine, a compound of biologic importance, with high precision, from about 7 K to over 300 K, and calculated the standard entropy of adenine. A list of values for several common substances is given in Table 7.7, and a more extensive one appears in Appendix 2A. One straightforward way is to combine standard enthalpy and entropy data from tables such as Tables 6.5 and 7.3. Note that while the majority of the values of standard enthalpies of formation are exothermic, or negative, there are a few compounds such as NO(g) and N 2O 4(g) that actually require energy from its surroundings during its formation these endothermic compounds are generally unstable.Entropy standard values, table Standard Gibbs free energies of formation can be determined in various ways. CompoundĪll values have units of kJ/mol and physical conditions of 298.15 K and 1 atm, referred to as the "standard state." These are the conditions under which values of standard enthalpies of formation are typically given. Table 1: Sample Table of Standard Enthalpy of Formation Values. Table 1 provides sample values of standard enthalpies of formation of various compounds. This is also the form with the lowest enthalpy, so graphite has a standard enthalpy of formation equal to zero. To determine which form is zero, the more stable form of carbon is chosen. The enthalpy difference between graphite and diamond is too large for both to have a standard enthalpy of formation of zero. The standard enthalpy of formation of a pure element is in its reference form its standard enthalpy formation is zero.Ĭarbon naturally exists as graphite and diamond. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
